Jarmo Aaltonen’s life changed in one fell swoop. After ice ball practice, his legs went out from under him in the locker room and Jarmo was rushed to Turku University Hospital. Cerebellar infarction. After that, many things took on new proportions and priorities changed.
“As a stroke patient, I was relatively young, in good general condition, so it came as a surprise,” Jarmo sums up.
It was December 2015 and immediately after Jarmo’s stroke he started taking anticoagulant warfarin. Warfarin is an atiocagulant that requires close monitoring and continuous measurement of INR values. Jarmo’s values did not remain at the desired level, so after six months the medication had to be changed to clopidogrel, which was continued for the next year and a half.
A stroke is no small thing. Jarmo was on sick leave for three months from his sales job and also banned from driving for a long time.
Jarmo obediently obeyed and changed his medication. When faced with such a significant issue, few patients find out the background of the active substance. My condition gradually started to improve and it was good to recover towards spring. Jarmo’s everyday life returned to normal and he felt lucky that he had overcome the fright.
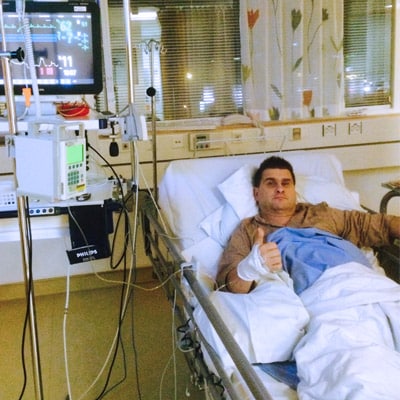
However, in the summer of 2017, the general condition began to slowly deteriorate, and by the autumn it had already deteriorated significantly. During the medication, the infarction did not recur, but later information on drug safety revolutionised the matter.
HIDDEN RISKS OF PROPHYLAXIS
‘During my hospital stay, I received literature about stroke and the risk of recurrence was discussed, but these things are not easy for someone who is not familiar with the subject,’ Aaltonen recalls.
Jarmo’s wife happened to hear about a pharmacogenetic test because of her own work. It is a test to find out everyone’s personal drug metabolism.
“Antithrombotic drugs came up and Plavix (clopidogrel) was mentioned. I asked my doctor the next day for a pharmacogenetic test. The doctor was amazed, but kindly sent a referral.”
“It was a relief. That I no longer had to eat unnecessary medicine that didn’t affect my body.”
A month after testing, the drug was changed. Jarmo had no response to Plavix. In practice, Jarmo had thought he was taking an anti-infarction drug, but tests showed that the drug did not work in Jarmo’s case. His body did not respond to clopidogrel, the active substance in Plavix. This information could only be determined by a pharmacogenetic test.
52,636 Finns are prescribed clopidogrel annually* (including the active substance in Plavix), 30% of whom do not have the desired effect. In other words, nearly 15,000 Finns eat the drug in question, unaware that it is useless or even harmful.
“It was a relief to know that I didn’t have to eat unnecessary medicine that didn’t affect my body” The medication was changed to another drug to which Jarmo’s genotype reacts. Side effects stopped after changing medication.
“There are many other drugs whose suitability is being investigated with this test.”
Fortunately, Jarmo was informed in time and the stroke has not returned.
*2017, KELA







